FDA action
We’re five years into GMPs (Good Manufacturing Practices) and FDA has noticeably stepped up enforcement in 2012.
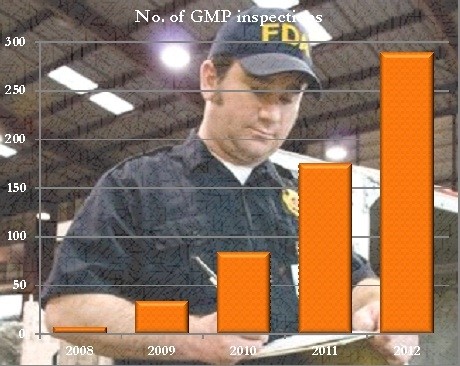
Only seven GMP inspections occurred in 2008, which increased to 34 in 2009, 84 in 2010, and 175 in 2011. FDA data as of September 1, 2012, showed that there had been 289 inspections in the program activity code.
GMP compliance has come under the spotlight, with an article in the Chicago Tribune in June claiming “serious and widespread manufacturing problems” among dietary supplement companies, with FDA officials reportedly citing violations in 50% of the 450 firms inspected over the last four years.
The quote that 50% of the industry was "failing on its face" represented a combination of OAI (Official Action Indicated) and VAI (Voluntary Action Indicated).
In an interview with NutraIngredients-USA at SupplySide West, FDA’s Dr Daniel Fabricant stood by his Tribune comments, and noted: “There is not a clear indication there will be a drop in non-compliance anytime soon, and that concerns us.”
2012 has also seen more injunctions and tougher actions from the agency. Dr Fabricant said that the industry will see a lot more strong GMP enforcement action in 2013.
“It sends a good message if we can take swift, effective, strong enforcement action. I hope it gives those people [who are flouting GMP regulations] a moment of pause.”
“I can’t emphasize greatly enough that I hope people move away from the mentality that FDA is just a traffic cop handing out tickets.”