Promotional Features
A new technology to counter iron deficiency
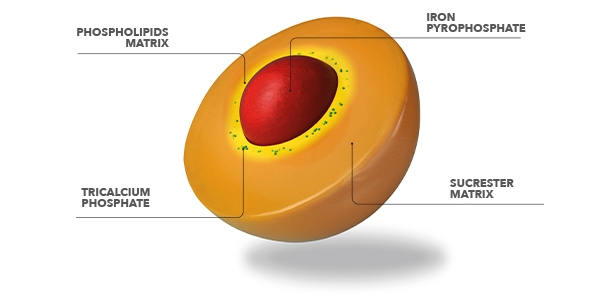
Sucrosomial iron is an innovative oral iron technology, in which ferric pyrophosphate (trivalent iron) is transported within a matrix of phospholipids and sucrose esters of fatty acids. This structure, called Sucrosome, allows high absorption of iron and optimal gastrointestinal tolerability.
Iron deficiency (ID)
Iron is an essential element for the body. It promotes the formation of hemoglobin and myoglobin, supports the immune system and normal cognitive function as well as supporting growth in children and adolescents.
The causes that can lead to the increase in the need for iron are different, but, in all cases, it is necessary to restore the body’s balance of this nutrient in order to facilitate certain normal physiological functions of our bodies.
Iron deficiency (ID) is a condition characterized by a low level of this mineral in the body, that, if not recovered, leads to an anemic clinical condition. Iron deficiency anemia (IDA) occurs when the body is not able to satisfy a right hemoglobin production, which is pivotal to ensure the oxygenation of tissues and cells of the body.
The main causes of ID are increased demand, reduced absorption, and increased loss of iron.
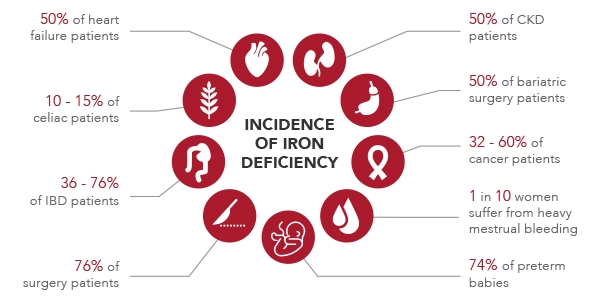
In the picture below is shown the presence of ID on specific clinical conditions:
Source: Susana Gómez-Ramírez et al., Sucrosomial Iron: A New Generation Iron for Improving Oral Supplementation.
World Health Organization (WHO) has defined the values of hemoglobin (Hb) as normal range at 14-18 g/dL for men and 12-16 g/dL for women; below these values, iron deficiency anemia is diagnosed. Other important markers to be considered for ID anemia are: ferritin, a storage protein able to contain up to 4500 iron atoms, transferrin saturation (%TSAT), as this glycoprotein is able to transport iron to the tissues and body districts that use this mineral, and sideremia, which indicates the amount of iron bound to transferrin in the bloodstream.
Iron Deficiency itself, instead, doesn’t always show hematological alterations; in fact, the hemoglobin level can be within the normal range, but the ferritin and the saturation of transferrin can be below. If the deficiency is not timely adjusted, it could develop in IDA.
The common symptoms related with ID and IDA, are:
- Paleness;
- Weakness;
- Dizziness;
- Irritability;
- Difficulty concentrating.
Commonly, ID is treated with oral iron salts (e.g. iron sulfate); however, conventional oral iron supplements are poorly absorbed by the body and lead to gastrointestinal side effects, with consequently reduced compliance and frequent drop outs by the patients.
Sucrosomial Iron (Sideral)
This new patented technology represents an innovative oral iron-containing carrier, in which ferric pyrophosphate is transported within a phospholipid plus sucrester matrix.
The result is a high gastrointestinal tolerability and easier iron absorption at the intestinal level. In the recent years many scientific evidences (both preclinical and clinical trials) have been published on International Journals confirming Sucrosomial technology effectiveness.
Gastro-resistance and Absorption
The gastro-resistance is due mainly to the sucrester matrix, as demonstrated by in vitro studies, that protects the iron from the acid gastric fluid.
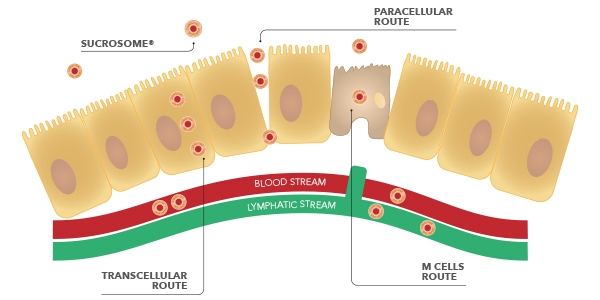
This allows the Sucrosome to entirely reach the intestinal mucosa, where it is absorbed by enterocytes through 2 routes: para-cellular and trans-cellular.
Besides, in the intestinal tract, also the Membranous cells (M cells) of the Peyer’s patches are involved for Sucrosomial iron absorption. Studies have demonstrated, in facts, that these cells are able to take part to the absorption of the Sucrosome and to transfer it to the lymphatic system.
Distribution
Scientific evidences on Sucrosomial iron demonstrate that this newer technology allows the absorption of iron by the intestinal tract and its distribution to the target tissues.
Considering its structure, similar to chylomicrons (gut lypoproteins able to transport lipophilic substances), it can be assumed that Sucrosomial iron reaches the liver, where the epatocytes are able to metabolize the phospholipid matrix, thanks to lysosomial enzymes, and to make available the ferric pyrophosphate for transferrin bound, and consequent tissues distribution, in particular to the bone marrow to form new red blood cells.
Iron Deficiency in Clinical Practice
Several studies in scientific literature confirm the efficacy of Sucrosomial iron supplementation in many clinical settings, where the deficiency of iron is frequent, for example: ginecology, oncology, nephrology, cardiology and especially in gastroenterology.
In gastroenterology, ID and IDA are the most frequent systemic complications in Inflammatory Bowel Disease (IBD), celiac disease and obesity. All these pathologies lead to chronic inflammation, that limits absorption and use of iron by the body. In patients with functional ID, the supplementation of conventional oral iron usually isn’t able to solve this problem, because proinflammatory cytokines are produced by the organism after inflammation. These cytokines stimulate the production and increase of hepcidin levels, a peptide hormone produced by the liver, which works by reducing serum iron concentration. Many studies in gastroenterology demonstrate that the supplementation with Sucrosomial iron allows to restore normal hematological ranges, and consequently to grant the normal function of red blood cells and hemoglobin.
Conclusion
The innovation of Sucrosomial technology is characterized by an excellent tolerability and it allows the iron intake anytime in the day (with meal or far from meals), for long periods of time, and prevents any discomfort commonly associated with iron intake, such as metallic and unpleasant aftertaste, irritation of the gastric mucosa, nausea or constipation. Overcoming the limits related to the conventional supplementation of iron, Sucrosomial iron promotes the intake of this important nutrient in all situations of deficiency or increased needs for iron.
Bibliography and Sitography:
- S. Gómez-Ramírez, E. Brilli, G. Tarantino, M. Muñoz. Sucrosomial Iron: A New Generation Iron for Improving Oral Supplementation.
Pharmaceuticals (Basel).2018 Oct 4;11(4):97.
- https://www.sideral.it/