The products, launched by Israeli company Ananada Scientific, builds on work done by Professor Nissim Garti at the Hebrew University of Jerusalem. Ananada launched the product at the recnet Marijuana Business Expo in Las Vegas under the Hygia Nutrients brand. The nano droplets of CBD are “are solubilized at the oil and water interface of the nano droplets, forming stable delivery vehicles loaded with the CBD,” according to the company.
Bioavailability question
Bioavailability of CBD (or cannabidiol, a non-narcotic fraction of Cannabis) has not been a big issue in the market in the past. But Ananada said Garti’s technology, known as the Lyotrophic Delivery System, was applied to CBD to get around a little publicized issue, that being that research has shown that some portion of CBD molecules are converted by the gastric juices of the stomach to THC, the narcotic fraction of Cannabis sativa. A 2007 study conducted in Japan found that a portion of CBD, when subjected to simulated gastric juices, converted to THC and two other cannabinoids, 9α-hydroxy- hexahydrocannabinol (9α-OH-HHC) and 8-hydroxy- iso-hexahydrocannabinol (8-OH-iso-HHC), that may have some degree of narcotic activity. Other studies, including this one from England, have noted the agonist effect CBD may have on the psychoactivity of THC, so the picture is complex meaning the psychoactivity of the ‘converted’ THC when CBD is present may be unnoticeable.
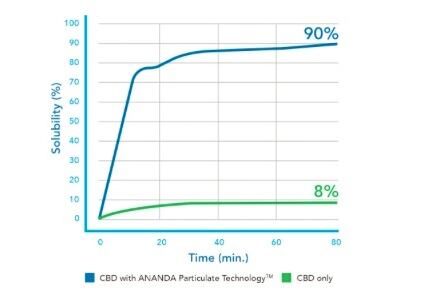
Nevertheless, Ananda claims that the issue has been insufficiently addressed in products currently on the market and also says that its own data has indicated this conversion actually happens at higher rates than those noted by the Japanese researchers.
“When CBD (even very pure) is taken orally (not by inhalation) it is transformed by the stomach acidic fluids to THC in a very short period of time. This is a known fact that was published by other scientists and was confirmed by us. After 30 minutes 70% of the CBD is transformed to THC. In our nano-droplets the CBD is protected against the gastric acidic fluids,” a company spokesperson told NutraIngredients-USA.
“The bioavailability of our products are much higher than the oil-based formulations (both faster to act and better to absorb) and as a results deliver better activity,” she added.
CBDs in the marketplace
The position of CBD products in the US marketplace is something of a gray area. Technically the whole plant is on the Drug Enforcement Administration’s schedule 1 list of controlled substances, meaning it is illegal at the federal level. But the federal government has adopted a hands-off attitude toward the trade in cannabis products and derivatives in those states that have adopted medicinal or recreational use laws. The Food and Drug Administration has ruled that CBD is not a legal dietary ingredient, and has based that ruling primarily on the fact that an English pharmaceutical company has begin to investigate it as a drug. But the benefits of CBD in the way it affects the body’s endocannabinoid system, an activity that could have implications for pain management, immune support and sugar metabolism and others, are so compelling that these hurdles have not kept product developers out of the space. It seems almost every month a new company pops up or a new product launch is announced.
One issue with CBDs has been the high cost of the extracts. This has to do as much with the distortions in the market caused by the unusual regulatory structure as it has to do with the difficulties of extraction itself. Costs are coming down, but in the meantime Ananada said this its LDS technology can help here, too .
“The formulation adds some cost but since so far the prices do not reflect the real production cost we will be able to decrease the price and also to get the same effect with smaller sizes,” the Ananada spokesperson said.
