Expert review: ‘DNA barcoding unsuitable as a stand-alone tool for identifying & authenticating botanicals’
DNA barcoding technology has rarely been far from the headlines since New York Attorney General Eric Schneiderman used it to build cases against a number of retailers of herbal supplements last year.
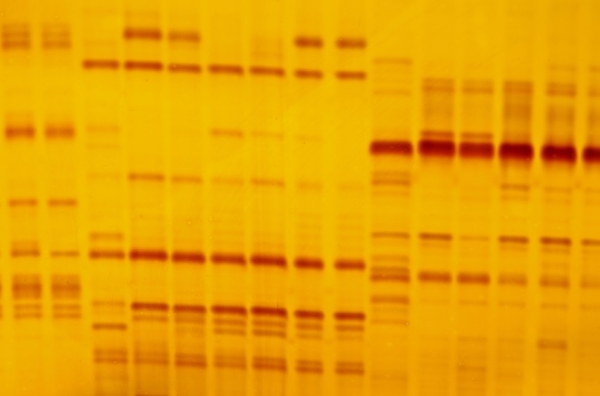
As reported by NutraIngredients-USA in 2013, DNA barcoding offers a lot of potential for botanical testing, and is incredibly reliable, but only when performed on appropriate material.
The new review, authored by scientists from the National Center for Natural Products Research at the University of Mississippi, the American Botanical Council, and the University of British Columbia, stated: “The major limitations to DNA barcoding of herbal products are related to the quality of DNA, primer affinity, PCR amplification, and sequencing of amplicons. Plant DNA is a relatively stable molecule and can be easily extracted from fresh or dried plant material using simple DNA extraction methods.
“However, the manufacturing process of herbal products that involves extensive heat treatment, irradiation, distillation, filtration, UV light exposure, and/or supercritical fluid extraction leads to either complete removal of DNA or degradation of DNA into smaller fragments. Hence, DNA barcoding is not feasible for processed herbal products such as extracts and tinctures in which the DNA is not present at all or highly degraded.”
The full paper is available as an open access article in Planta Medica.
"If we could turn back the clock..."

Mark Blumenthal, Founder & Executive Director of the American Botanical Council, told us: "As is the case in previous (and future) publications, ABC is pleased to have a partnership with the National Center for Natural Products Research at the University of Mississippi in which we are committed to educating members of the scientific community, botanical industry, and others about the rational ways to optimize the laboratory analyses of botanical raw materials and extracts, as well as finished dietary supplement products.
"With respect to the much-misunderstood role of DNA barcoding analytical techniques in the identification of botanical materials and extracts, this new paper, extensively peer reviewed and published in one of the most respected medicinal plant research journals, helps to clarify the strengths and weaknesses — the appropriate and inappropriate uses — of DNA barcoding techniques for botanical materials. The conclusions of this paper are consistent with previous publications by experts in DNA-based technologies and other analytical methods for botanicals.
"If we could turn the clock back to pre-February 2015, and if this highly authoritative paper were available in the scientific literature at that time, perhaps the New York Attorney General would not have taken his misguided and scientifically-flawed actions against the four retailers whose herbal dietary supplements the AG had tested using only a DNA method which apparently was ill-suited and inadequate for such testing."
Scientific consensus
The review’s conclusions echo results presented by scientists from the US Food and Drug Administration and an Australian research consortium at the prestigious 16th Annual Oxford International Conference on the Science of Botanicals at the University of Mississippi in April.
Commenting on the results presented at the Oxford conference in April, a spokesperson for NY AG’s office emailed the following statement: “We believe that DNA barcoding is a critical part of improving the industry’s approach to ensuring identity and avoiding contamination.
“We’re heartened by the responsible voices in the supplements industry who agree—and are beginning to incorporate DNA barcoding as part of their testing protocols.”
When contacted about the new review this week the NY AG’s office declined to comment.
Strengths & limitations of DNA barcoding
Top level takeaways from the new review include:
- DNA barcoding is more successful if carried out on raw herbal materials before processing;
- It is also more successful when carried out on dried or powdered raw material versus the final extract or tincture;
- DNA barcoding cannot differentiate between plant parts (root versus leaf, for example), nor does it differentiate between plants within the same species;
- Because excipients/ fillers are added after processing of the herbal ingredient, DNA barcoding could preferentially amplify DNA from excipients, and possibly give a false negative result for the herbal species (this could be overcome by next gen sequencing techniques);
- There is a lack of reference libraries and voucher specimens for the DNA sequences referenced in the GenBank database.
“To overcome these limitations of DNA-based analysis, chemical profiling or other analytical chemistry methods should be adopted for authentication of herbal products. Thus, DNA barcoding should go hand-in-hand with chemical analysis and macroscopic and/or microscopic evaluation to tackle the adulteration problems prevailing in the herbal industry,” wrote the authors.
“The creation of a mini-barcode reference library or Herb-BOL, containing all of the authentic reference barcode sequences linked to the respective taxonomically validated herbarium vouchers, would be a useful tool to ensure access to reliable DNA barcodes. The use of a barcode reference library could provide a basis for using DNA technologies as a cGMP-compliant approach for the authentication of herbal products and dietary supplements in the future.”
A powerful tool, but...
Corey Hilmas, MD, PhD, Sr VP of Scientific & Regulatory Affairs at the Natural Products Association (NPA), told us: "We agree with the authors conclusions for DNA barcoding as unsuitable as a stand-alone tool for identifying and authenticating the herbal plant species. DNA barcoding does not address the part of the plant used in the manufacture of the product, and there are inherent difficulties with highly processed botanical ingredients used in dietary supplements because it may not contain any usable DNA long enough to amplify with PCR [polymerase chain reaction] for the chosen DNA barcode.
"Nevertheless, DNA barcoding is a powerful tool that can be used with other methods like chemical fingerprinting. NPA reached this conclusion with our joint press release with the NY AG over one year ago in May 2015. We stated 'While no single test or technology alone can provide complete confidence to consumers, we support the application of DNA barcoding technology as part of a multifaceted approach to assuring authenticity and identifying substitution.' I think that is the same conclusion and take away message the authors of this article came away with."
NPA published a white paper on 'DNA Barcoding for Botanical Authentication' in March 2015. To access the white paper, please click HERE.
Source: Planta Medica
Published online ahead of print, doi: 10.1055/s-0042-111208
“DNA Barcoding for the Identification of Botanicals in Herbal Medicine and Dietary Supplements: Strengths and Limitations”
Authors: I. Parveen et al.