The explosion of research into the role the gut microbiota plays in human health has created a wealth of opportunities to use probiotics to maintain and improve wellbeing. However, the growth in knowledge of the links between microorganisms and health outcomes has outpaced understanding of how externally introduced probiotics affect the microbiome and, crucially, whether they have the desired effects. In light of the knowledge gap, Morinaga Milk Industry has worked to understand a widely used probiotic strain.
There is now a large body of evidence linking the gut microbiome to human health and disease. Studies have implicated changes in microbial communities and the balance of host-microbiota interactions to a wide range of conditions, including inflammatory bowel disease, Type 2 diabetes, and obesity.1 The work has spurred further research suggesting that the use of probiotics to modulate the gut microbiota affects the health of the host.2 Researchers have reported prophylactic and therapeutic effects on health.3
Despite years of research, there remains a need for a more detailed picture of the effects of probiotics on the gut microbiome and, by extension, human health. There is now a pool of probiotics, each with its own mechanism of action, safety profile, origin, and efficacy. Those differences matter. Studies show that the positive effects of probiotics can be specific to particular strains.4
Nutrition companies need to understand the strain-specific effects of probiotics to ensure their products have the desired impact on human health. Yet, there is a lack of in vitro and human data on many of the available probiotic strains.
Bifidobacterium longum subsp. longum BB536 shows it is possible to build up a detailed understanding of a probiotic strain and its effects on the microbiome. Since its discovery decades ago, B. longum BB536 has been used as a probiotic and studied to show how and why it affects human health and wellbeing. To support informed decisions about probiotic selection, Morinaga Milk published a summary of the effects of the commercially available human-residential bifidobacteria (HRB) in the Journal of Functional Foods.5
Manufacturers have incorporated BB536 into milk-based drinks, yogurts, infant formula, and nutritional supplements since its discovery in the gut of a healthy breast-fed infant in 1969. Throughout the use of the strain, and particularly over the past decade, researchers have sought to shed light on what happens when people consume BB536.
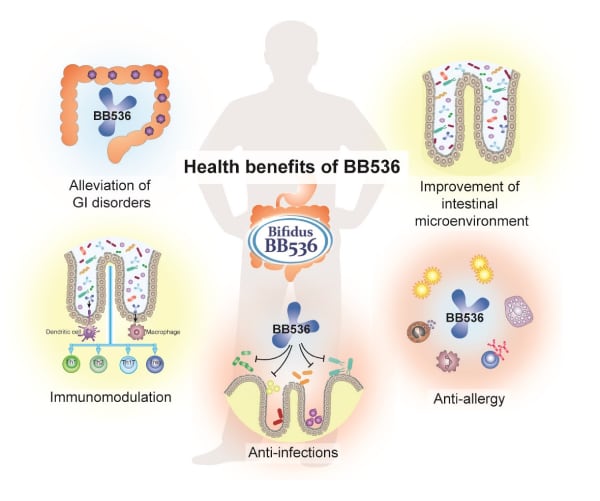
How BB536 modulates the microbiome
Research suggests BB536 supports gut homeostasis. When people consume an animal-based diet, their microbial profile changes, with the proportion of bile-tolerant bacteria rising at the expense of the Firmicutes that metabolize dietary plant polysaccharide. Researchers have linked increased abundance of the bile-tolerant bacteria Bilophila wadsworthia to the development of inflammatory bowel disease and colorectal cancer.6–8
BB536 may be able to counter changes caused by animal-based diets. A randomized clinical trial found consumption of yogurt supplemented with BB536 maintained a normal microbiota composition when eating an animal-based diet.9 The finding points to the ability of the strain to stabilize the gut microbiota, improve the intestinal environment, and enhance gut health.
Stabilization of the gut microbiota is part of a broader range of changes triggered by BB536 that indicate how it affects human health. Researchers have shown how the probiotic interacts and cross-feeds with endogenous gut microbiota, promotes beneficial metabolite production in the microbiome, and helps to alleviate allergic diseases.
The evidence for cross-feeding of other species of human gut-derived microbiota comes from studies of the levels of intestinal butyrate and the prevalence of butyrate-producing Eubacterium rectale.10 Studies have shown BB536 makes acetate through the fermentation of carbohydrates, thereby stimulating the growth of butyrate-producing colon bacteria and the production of butyrate.11 The work is part of a body of evidence suggesting BB536 improves gut homeostasis through cross-talk with other bacteria.
There is evidence that the modulation of the gut microbiome by BB536 has anti-allergy effects. A large collection of studies have looked at the effect of BB536 on Japanese cedar pollinosis, an allergy triggered by exposure to certain pollen, and other conditions such as pollen-induced allergic rhinitis.12,13 The work found the probiotic may protect against allergic reactions and improve quality of life.
Collectively, the research summarized by Morinaga Milk indicates that B. longum BB536 stabilizes the gut microbiota and interacts with other microorganisms to have beneficial effects. The years of research are enabling manufacturers to create probiotic products backed by evidence of strain-specific effects on the gut microbiota and human health.

References
1. Liang, D., Leung, R. K.-K., Guan, W. & Au, W. W. Involvement of gut microbiome in human health and disease: brief overview, knowledge gaps and research opportunities. Gut Pathog. 10, 1–9 (2018).
2. Azad, M. A. K., Sarker, M., Li, T. & Yin, J. Probiotic Species in the Modulation of Gut Microbiota: An Overview. Biomed Res. Int. 2018, (2018).
3. Scott, K. P., Antoine, J.-M., Midtvedt, T. & van Hemert, S. Manipulating the gut microbiota to maintain health and treat disease. Microb. Ecol. Health Dis. 26, 25877 (2015).
4. McFarland, L. V., Evans, C. T. & Goldstein, E. J. C. Strain-Specificity and Disease-Specificity of Probiotic Efficacy: A Systematic Review and Meta-Analysis. Frontiers in Medicine 5, 124 (2018).
5. Wong, C. B., Odamaki, T. & Xiao, J.-Z. Beneficial effects of Bifidobacterium longum subsp. longum BB536 on human health: Modulation of gut microbiome as the principal action. J. Funct. Foods 54, 506–519 (2019).
6. Devkota, S. et al. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10-/- mice. Nature 487, 104–108 (2012).
7. Han, S. et al. Intestinal microorganisms involved in colorectal cancer complicated with dyslipidosis. Cancer Biol. Ther. 20, 81–89 (2019).
8. Yazici, C. et al. Bilophila wadsworthia Is More Abundant in the Colonic Microbiome of Colorectal Cancer Cases Compared to Healthy Controls. Gastroenterology 148, S–100 (2015).
9. Odamaki, T. et al. Effect of probiotic yoghurt on animal-based diet-induced change in gut microbiota: an open, randomised, parallel-group study. Benef. Microbes 7, 473–484 (2016).
10. Sugahara, H. et al. Probiotic Bifidobacterium longum alters gut luminal metabolism through modification of the gut microbial community. Sci. Rep. 5, 13548 (2015).
11. Falony, G., Vlachou, A., Verbrugghe, K. & De Vuyst, L. Cross-feeding between Bifidobacterium longum BB536 and acetate-converting, butyrate-producing colon bacteria during growth on oligofructose. Appl. Environ. Microbiol. 72, 7835–7841 (2006).
12. Xiao, J.-Z. et al. Clinical efficacy of probiotic Bifidobacterium longum for the treatment of symptoms of Japanese cedar pollen allergy in subjects evaluated in an environmental exposure unit. Allergol. Int. 56, 67–75 (2007).
13. Miraglia Del Giudice, M. et al. Bifidobacterium mixture (B longum BB536, B infantis M-63, B breve M-16V) treatment in children with seasonal allergic rhinitis and intermittent asthma. Ital. J. Pediatr. 43, 25 (2017).








