Promotional Features
Pantesin® pantethine: Comprehensively aiding cardiovascular health for 40 years
Cardiovascular disease represents a major opportunity for manufacturers of nutritional supplements to improve health. More than 7 million people a year die of heart attacks. Through preventative steps to lower LDL cholesterol and affect the levels of other molecules, some of these deaths could be avoided. In Pantesin® pantethine, the nutritional industry has access to an ingredient with a 40-year track record of safely lowering LDL cholesterol and the levels of other molecules.
Given the widespread nature of cardiovascular disease, the potential to improve health is significant. Heart attacks kill 370,000 people a year in the US and 20 times as many people worldwide. Research suggests many of these deaths could be prevented by lowering cholesterol. The risk calculation is straightforward. For every 1% decline in LDL cholesterol, the relative risk of suffering a major coronary heart disease event, namely a heart attack, falls by 1%. As such, a worldwide 1% drop in levels of LDL cholesterol is predicted to result in 73,000 fewer deaths from heart attacks each year.
People at high risk of suffering from cardiovascular disease can take statins to bring their levels of LDL cholesterol down toward the target value. Yet for people at low to moderate risk of cardiovascular disease, such interventions are unnecessary and potentially counterproductive given the risk of side effects from long-term use and evidence suggesting it is harmful to lower LDL levels by too much. This large population of people who want to control their levels of LDL cholesterol and other molecules but do not need statins require products to help them achieve their health goals.
Extensive research into Pantesin® pantethine spanning four decades suggests the ingredient can play a role in this process. In vitro tests have demonstrated pantethine, a derivative of pantothenic acid and precursor of coenzyme A (CoA), inhibits the synthesis of cholesterol and fatty acids by 80% and 50%, respectively. Pantethine drives such inhibition by increasing the availability of CoA, which, in turn, enhances the Krebs cycle and cuts the use of acetate for the synthesis of fatty acids and cholesterol. Importantly, data from studies in humans validate the real-world impact of this mechanism of action.
A comprehensive effect on cardiovascular risk factors
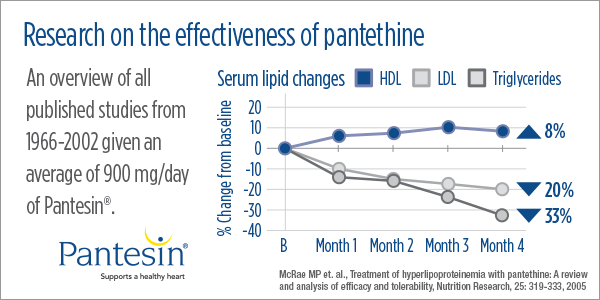
The 40-year history of pantethine use in Japan has resulted in the creation of a significant body of evidence to support the safety and efficacy of the ingredient. An overwhelming majority of the trials, particularly in healthy subjects, have tested Pantesin®, a pharmaceutical-grade pantethine brand sold by Kyowa Hakko. The scale and scope of the research is evident in a review Marc McRae, a professor at the Department of Physiology and Biochemistry, National University of Health Sciences, published in Nutrition Research in 2004.
McRae’s paper identified 28 clinical trials of pantethine through a literature search spanning from January 1966 to December 2002. In total, the trials enrolled 646 subjects. Collectively, the 28 clinical trials found four months of treatment with pantethine resulted in a 20.1% reduction in LDL cholesterol. This equates to a 20% reduction in the relative risk of a major coronary heart disease event. Notably, the ingredient was also associated with a positive effect on other molecules associated with cardiovascular health.
Following four months of treatment with pantethine, participants on average experienced a 32.9% decrease in levels of triacylglycerols, combinations of three fatty acids and one glycerol molecule also known as triglycerides. Studies have linked triglycerides to cardiovascular health. “The evidence that raised concentrations of remnant cholesterol, marked by raised triglycerides, are an additional causal risk factor for cardiovascular disease and all-cause mortality, is increasing,” researchers wrote in The Lancet in 2014.
The McRae paper also suggested pantethine can affect levels of HDL, which is commonly known as the ‘good’ cholesterol. Across the 28 studies, pantethine was associated with a 10.7% increase in HDL levels after three months. While the data are less compelling than for triacylglycerols and LDL, they add to evidence in support of the comprehensive effect pantethine has on cardiovascular health. This is significant when the effects of pantethine are considered in light of data in support of the use of alternative ingredients.
While omega-3 is primarily associated with the lowering of triglyceride levels and sterols are typically taken to reduce LDL cholesterol, the McRae review suggests pantethine acts on multiple markers of cardiovascular health simultaneously. As suboptimal levels of LDL, HDL and triglycerides have all been associated with a higher risk of cardiovascular disease, there appears to be value in taking a nutritional supplement that has a positive effect on each of the markers, rather than a product that only modulates the levels of one molecule.
Significant improvements in healthy subjects
The steep increases and decreases in LDL, HDL and triglycerides identified in the McRae paper were seen in trials that enrolled some subjects that had unhealthy levels of the markers going into the study. In healthcare today, such high-risk patients are catered for by statins and other prescription drugs. The Third Report of the National Cholesterol Education Program Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (ATP III) established a clinical paradigm for when to start a patient on statins.
In doing so, ATP III, and the 2004 revision ATP IV, created a subpopulation of patients who have levels of LDL cholesterol that are abnormal, but no so abnormal that they are eligible for treatment with statins. The ATP III recommended people in this subpopulation start the therapeutic lifestyle change (TLC) diet, a program that encourages individuals to revise their eating, exercise and smoking habits in a bid to lower their levels of LDL cholesterol. Researchers have subsequently run trials to assess the potential for supplementation with Pantesin pantethine to support the TLC diet program.
Dr John Rumberger, the Director of Cardiac Imaging and Lipid Management at Princeton Longevity Center, published the results of such a study in Nutrition Research in 2011. The clinical trial enrolled 120 people in North America with low to moderate risk of cardiovascular disease and started them on the TLC diet. After four weeks on the TLC diet, participants were randomized to either Pantesin or placebo. The treatment arm took 600 mg daily doses of Pantesin pantethine in the first half of the 16-week study, and 900 mg in the second half. All subjects continued the diet throughout the study.
Sustained, statistically-significant declines in the levels of markers of cardiovascular risk were seen in the treatment arm. After 16 weeks, total cholesterol was down 3%, LDL cholesterol was down 4% and apolipoprotein B was down 5%, results that show Pantesin pantethine can drive reductions above and beyond what can be achieved through diet alone. As expected, the percentage declines were lower than were seen in the McRae review. The discrepancy is a result of the health of the subjects going into the trials.
In the Dr Rumberger study, all potential subjects who were found to be at high risk of cardiovascular disease according to the ATP-III categories were referred to their primary care provider for follow up. As such, all of the participants had levels of LDL cholesterol that were just above normal. In contrast, the McRae review included historic clinical trials that enrolled people with higher starting levels of LDL cholesterol. These historic studies linked Pantesin pantethine to larger percentage declines in LDL cholesterol because the participants were further from normal going into the trial.
This hypothesis is supported by a subsequent analysis of the moderate-risk subgroup of patients in the Dr Rumberger study, details of which were published in Vascular Health and Risk Management in 2014. Among these higher-risk patients, Pantesin pantethine drove an 11% decrease in levels of LDL cholesterol. In contrast, participants in the placebo arm, who were on a diet designed to lower their cholesterol, experienced a 3% increase in LDL over the course of the study. Similar trends were seen in levels of total cholesterol and apolipoprotein B.
A safe, effective way to lower cholesterol
Collectively, the McRae review and two Dr Rumberger papers show that the scale of the effect of Pantesin on LDL cholesterol and other markers is dependent on the health of participants at the start of treatment. Less healthy patients experience larger changes. In today’s treatment paradigm, the effect on low to moderate-risk patients is of most real-world significance. The 11% decline in LDL cholesterol in moderate-risk patients corresponds to an 11% drop in risk of major coronary event. In a condition as prevalent as cardiovascular disease, even a 4% decline affects a lot of people.
Notably, low to moderate-risk patients can reduce their LDL without affecting their near-term health. Among the 646 participants analyzed in McRae’s review, only one person withdrew because of a treatment-related side effect. The adverse event, gastric discomfort, is experienced whenever people start a new treatment. In the case of Pantesin pantethine, it clears up spontaneously and typically within the relatively short durations of the clinical trials. When paired to 40 years of data on the real-world use of Pantesin pantethine, this amounts to a favorable safety and tolerability profile.
The safety profile is one of the two main ways Pantesin pantethine stands out from the competition. The other is its comprehensive effect on markers of cardiovascular health, a characteristic backed up by decades of research into how levels of LDL cholesterol, triglycerides and other molecules change following treatment with Pantesin pantethine.