Lack of regulatory ‘middle ground’ keeps Echinacea from shining in the US, says manufacturer

But despite its high popularity ranking, the category still has a lot more room for stateside growth, according to Pierce Sioussat, CEO of Bioforce, a New York-based company that distributes finished products by European supplement brands.

“From a regulatory perspective, there are a lot of claims [dietary supplements] can’t make even though the research says those claims may be valid,” he told NutraIngredients-USA.
His company distributes the Echinaforce line by Swiss natural product manufacturer A. Vogel in the US.
“You have your pharmaceuticals follow one path, at the other end of the spectrum you have products that follow DSHEA protocols—but there’s nothing really in the middle where you can take an herbal medicine that has some clinical substantiation for products that are involved, then you can communicate very clearly to consumers what those products are.”
Regulatory structure reflected in Echinacea’s flat sales
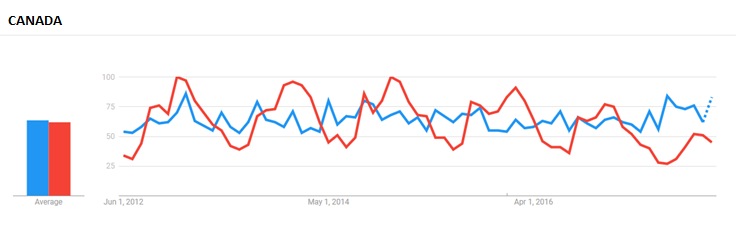
For comparison, NutraIngredients-USA looked at Google searches for Echinacea and the phrase ‘Cold Remedies’ in the past five years. Worldwide, searches for Echinacea peaked around the same time as searches for ‘cold remedies’ (in December, around ‘cold season’ for most of the Northern Hemisphere).
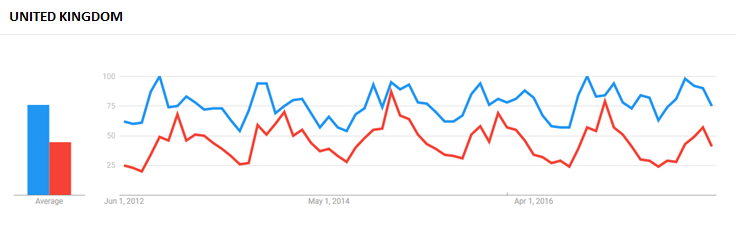
Then we compared results in the English-speaking countries of Canada, the UK, and the US. In the first two countries, Echinacea is registered as a health ingredient by Health Canada and the MHRA, supporting the use of common cold-related claims.
BLUE: Echinacea, RED: Cold Remedies
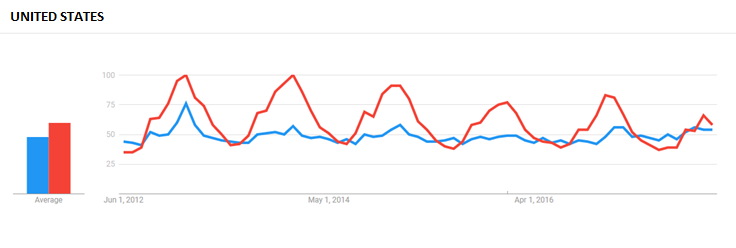
In the UK, which Sioussat said has a much more mature Echinacea market among the markets compared, searches for Echinacea outpaced that for ‘cold remedies.’ The distribution was more random in searches by Canadian internet users, while searches for Echinacea in the US trailed off and remained steady in the period between 2012 and 2016.
Diversity of Echinacea varieties, preparations, a drawback for marketing and research
“I think that echinacea has a developing and growing clinical body of evidence for supporting its use for helping to prevent and possibly treat upper respiratory tract infections related to colds and flu. But one of the big problems with echinacea in the clinical literature is that there are many different preparations that have been used generically,” Mark Blumenthal, founder and executive director of the American Botanical Council, told NutraIngredients-USA. READ MORE
US sales data provided by market research firm SPINS mirrors American search patterns for Echinacea. Combining all sales channels, total sales of products containing Echinacea in the vitamins and supplements category have declined year-over-year since SPINS started tracking it in 2014, -52% in November 2015, -65% in November 2016, and -83% in November 2017.
Stronghold in the natural channel
But when SPINS data was isolated to reflect sales in the natural channel—a stronghold for US sales of Echinacea products—it shows steady increases.
Sioussat said that the natural channel has been the easiest to penetrate with the Echinaforce product. Still, the product variety offered in the US isn’t as diverse as in Canada or the UK.
Mirroring public awareness of Echinacea and how the ingredient is regulated in each market, Bioforce is not marketing as many Echinaforce products under the A. Vogel brand in the US compared to what is sold in Canada and the UK.
“What we have in the US right now is the liquid extract, and we have the caplets,” he said. Meanwhile, in Canada and the UK, Echinaforce also comes in lozenges and a throat spray format. Sioussat added that he anticipates the Echinacea-Sage throat spray product to hit US shelves next year, slated for an introduction at Expo East 2018.
To reignite Echinacea sales and awareness in the US, Sioussat said that the company is putting more focus on working with retailers for in-store education. That’s one reason the natural channel has been more successful.
“In the natural channel, you have retailers which are much more engaged,” he said. “You walk into a [big box store], you’re not going to get that.”