The Centers for Disease Control and Prevention (CDC) said 87 people are ill from 35 states including 27 hospitalisations.
CDC is recommending against consuming any kratom because no common brands or suppliers have been identified.
Forty of 55 people interviewed reported consuming kratom in pills, powder or tea. Most people had the powder form. The majority report having used Maeng da red vein kratom.
Dave Herman, American Kratom Association chairman of the board, previously said the FDA is ‘going against sound science’ when it comes to kratom.
Three strains added to investigation
The Food and Drug Administration (FDA) said Salmonella Javiana, Salmonella Okatie and Salmonella Thompson are now part of the investigation as well as the original strain, Salmonella I 4,[5],12:b:-.
FDA said 25 product samples have tested positive for Salmonella. Three revealed isolates genetically related to an outbreak strain, while 22 were positive for a non-matching Salmonella or Salmonella and further testing is pending.
Salmonella I 4,[5],12:b:- has caused 50 illnesses, S. Javiana (five), S. Okatie (16) and S. Thompson (16).
CDC said the same strains of S. Okatie and S. Thompson were found in samples of kratom and those from ill people.
Illness dates range from 21 January 2017 to 24 February 2018. Sick people are aged six to 67 and 55% are male.
States find contaminated kratom
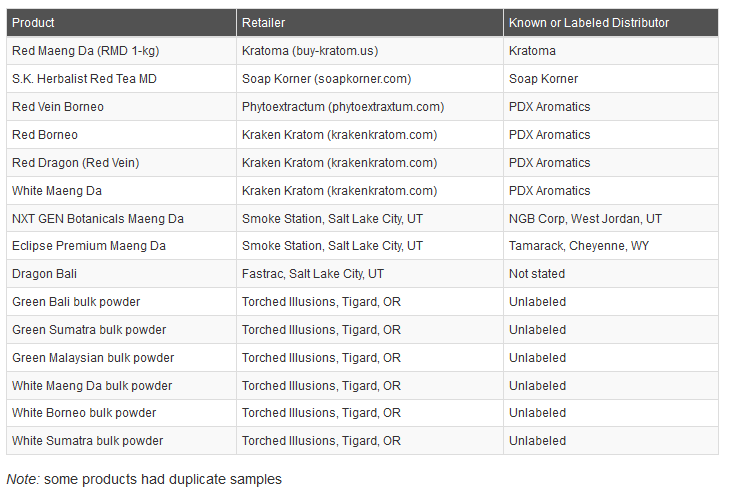
PDX Aromatics, doing business as Kraken Kratom, Phytoextractum and Soul Speciosa, recalled kratom-containing powder products after testing by the California Department of Public Health identified Salmonella in an open product sample.
FDA testing confirmed presence of the pathogen in products from the company.
Investigators in Oregon and Utah collected kratom powder from retail and online retailers where ill people purchased the supplement. Outbreak strains of S. Okatie and S. Thompson were found.
The ill person in Utah bought kratom powder from online retailer Kratoma.
Testing by North Dakota Department of Health identified Salmonella I 4,[5],12:b:- in opened, leftover kratom powder collected from ill people.
The infected person purchased S.K. Herbalist brand kratom powder from online retailer Soap Korner.
“Additional testing by states has revealed Salmonella in other brands of kratom, meaning it is likely that multiple kratom and kratom-containing brands and retailers are supplying contaminated product to the public,” said FDA.
“The positive results also indicate possible concerns with the manufacturing practices used in production and/or handling of these products.”
The agency added there is no FDA-approved use for kratom.
Meanwhile, CDC has launched an online tool with access to nearly 20 years of data on foodborne, waterborne and other enteric (intestinal) disease outbreaks.
Information in the NORS Dashboard comes from the National Outbreak Reporting System (NORS).
It replaces the Foodborne Outbreak Online Database (FOOD Tool) which provided data only on foodborne outbreaks in NORS.
