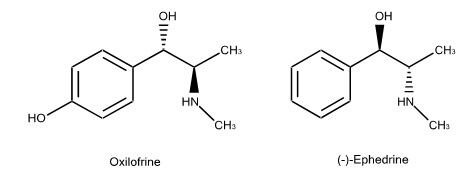
Methylsynephrine is also known as oxilofrine and p-hydroxyephedrine, and is not a legal dietary ingredient. The US Food and Drug Administration sent warning letters dated March 31, 2016 to 7 companies regarding a total of 8 products marketed as dietary supplements and containing methylsynephrine as a dietary ingredient.
New data published in Drug Testing & Analysis indicated that, of the 27 products listing methylsynephrine on the label, 14 actually contained the ingredient. Doses ranged from 0.0003 to 75mg per individual serving.
If consumers followed the instructions on the label they could ingest 250mg of methylsynephrine per day. Typical prescription doses for oxilofrine range from 16 to 40mg for the stimulation of the heart and to increase blood pressure.
“The potential adverse health effects of inadvertently consuming up to 250mg of oxilofrine per day, as would be possible with supplements in our study, are entirely unknown,” wrote the authors. “Human studies using lower dosages suggest that the increased demand on the heart muscles might lead to palpitations, arrhythmias, increased blood pressure, or other adverse effects.
“Of particular concern with respect to the safety of oxilofrine in sports supplements is that more than one million youth athletes in the USA use sports supplements. Their relatively small developing bodies might be particularly vulnerable to adult dosages of oxilofrine. Usual oxilofrine doses for children and adolescents range from 8 to 24 mg; therefore, adolescents using supplements analyzed in our study might consume more than 3 times the highest pediatric dose of oxilofrine.”
The study was performed by scientists from Harvard Medical School, Cambridge Health Alliance, the National Center for Natural Products Research at the University of Mississippi, the Dutch National Institute for Public Health and the Environment, and NSF International.
John Travis, NSF International Research Scientist and co-author on the paper, said: "As a public health organization we want to warn consumers about these compounds that may harm instead of improve their health.
"The presence of potentially harmful compounds such as oxilofrine, disguised as ‘methylsynephrine’ or ‘extract of Acacia rigidula,’ in over-the-counter supplements is illegal, and consumers should be aware of and avoid products containing these compounds. NSF International developed a testing and certification program that verifies supplement labels are accurate and the product itself is free from harmful levels of contaminants such as oxilofrine. We urge consumers to seek NSF certified dietary supplements to avoid unintentionally consuming harmful compounds."
Troubling and disturbing
Steve Mister, president and CEO of the Council for Responsible Nutrition (CRN), called the study’s findings “particularly troubling” to responsible marketers of dietary supplements “because they demonstrate once again that rogue companies will ignore the laws designed to protect consumers.
“Oxilofrine is not a legitimate dietary ingredient, and its marketers have not complied with the legal requirements for bringing a new dietary ingredient to market so it cannot legally be sold in dietary supplements,” he said.
“We appreciate the work that Dr Cohen and the co-authors of this new study have done, and we hope this will result in isolating fringe players that recklessly break the law. As the authors acknowledge, ‘the US law regulating supplements does not permit dietary supplements to contain unapproved pharmaceutical drugs such as oxilofrine.’”
Mister commended FDA’s enforcement action against seven companies marketing products containing methylsynephrine/oxilofrine. “We urge FDA to use all the resources at its disposal to take action to sanction these companies—and others—to remove the products from the marketplace. We urge consumers to follow FDA’s advice and not use any products marketed as supplements that contain methylsynephrine, oxilofrine, or p-hydroxyephedrine.”
“This type of blatant illegal activity is not only disturbing, but also disruptive, to companies in the dietary supplement industry who sell reputable products,” added Mister. “The sports nutrition sector is one of the fastest growing sectors in the industry and there are a lot of legitimate products in that space. We need to be careful because we don’t want to lose the customers in that space, and so FDA needs to move quickly to remove illegal products from shelves. If we have a serious health event it could kill the category.”
Source: Drug Testing & Analysis
Published online, doi: 10.1002/dta.1976
“Pharmaceutical doses of the banned stimulant oxilofrine found in dietary supplements sold in the USA”
Authors: P.A. Cohen et al.