News in Brief
CBD earmark advocated by NPA makes it into final House version of budget bill

NPA announced today that the amendment, sponsored by Rep. Jerry McNerney (D-CA), was included in the House Appropriations Bill that was approved by a slim majority of the full chamber. The bill now goes to the US Senate.
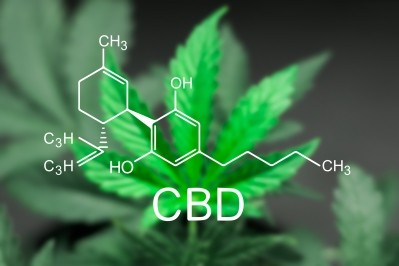
McNerney’s amendment sets as $100,000 for FDA to study the issue of what would be an appropriate upper limit for CBD daily exposure. It’s a critical question, as the molecule, either as an isolate or as part of a ‘full spectrum’ hemp extract, is showing up in products of all sorts, including topicals, softgels, tinctures and foods.
There are as many as 1,500 of these products currently on the market, with widely varying CBD dosages. So it’s an open question as to how much a consumer who’s enthusiastic about these types of products might be ingesting in a day.
Having the amendment in the final version of the House bill is the good news. The not so good news, according to NPA president and CEO Dan Fabricant, PhD, is that the House bill is as much as $60 billion over the amount previously agreed to between the two chambers. So there is a still a lot of horse trading to be done.
But Fabricant said that just having the language on CBD included in a version of the bill is a big step forward.
“With all of the political mumbo jumbo, including talk about a possible impeachment of the President, it’s unlikely that this is the final word on the budget,” Fabricant said. “But this puts it in play.”