Proposed changes to supplement labels could cause many products to be reformulated

The new labeling regime will entail some expense on the part of dietary supplement companies, but firms will have plenty of time to prepare, said attorney Justin Prochnow.
Long time to prepare
“Right now we’re in the 90-day comment period and I would be very surprised if that comment period is not extended another 90 days,” Prochnow, a shareholder in the firm Greenberg Traurig, told NutraIngredients-USA. “Then it would take them some time to go through the comments. The very earliest they would issue a final rule would be two to two and a half years down the road, but we are probably looking at something more like three or three and a half years.”
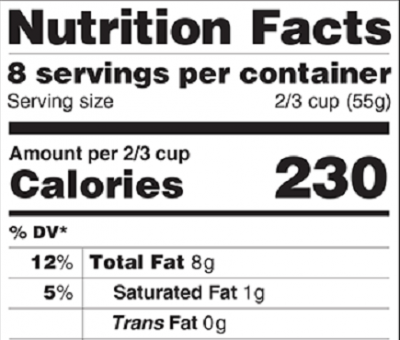
Prochnow said the changes for supplement facts labels are much less pronounced than for the nutrition facts panel (for a discussion of those changes, click here. To read FDA's full draft proposal, click here.)
“It’s a pretty stark contrast from nutrition facts panels. The supplement facts panels don’t change that much. It’s mostly the proposed changes in daily values, and there are some pretty significant differences,” he said.
The new labels will up the emphasis on vitamin D and potassium, adding those two nutrients to calcium and iron among mandatory nutrient disclosues on food labels. FDA says that the vast majority of the population is deficient in vitamin D.
The changing levels of nutrients, based on more recent science, may necessitate reformuation of many products, FDA admits. And Prochnow said manufacturers may have to reformulate if they want to continue to make compliant nutrient content claims, such as “good souce of” or “excellent source of.” These are based on the product having a specified percentage of the RDI for a given nutrient. If a company was putting just enough of a given nutrient to quailify for a nutrient content claim under the old rules (as many companies were doing), more of that material will need to be put into a product to qualify in the future, Prochnow said.
Update needed
Regardless of what the costs may be to industry, the update was long overdue in the eyes of the Council for Responsible Nutrition.
“The current daily values are based on evidence from 1968 and clearly there has been quite a bit of progress on nutritional values since then,” said Andrea Wong, PhD, vice president of scienfitic and regulatory affairs for CRN.
“We definitely commend the efforts of the agency and the adminstration in recognizing that Recommended Daily Allowance is the most appropriate measure for estabilshing the Reference Dietary Intake,” she said. “At a time when we know of significant nutrient shortfalls in the typical American diet, it would have been a mistake to water down nutritional requirements so that at most only half the population would be covered,” Wong said.
The RDA is the daily dietary intake level of a nutrient considered sufficient by the Food and Nutrition Board to meet the requirements of 97.5% of healthy individuals in each life-stage and sex group. It is calculated based on the Estimated Average Requirements (EAR), and is usually approximately 20% higher than the EAR. The EAR is expected to satisfy the needs of only 50% of the people in that age group based on a review of the scientific literature.
Wong also said CRN was pleased with FDA’s discussion of the criticism of supplementation in some quarters, namely that using supplements, given the widespread practice of fortifying certain foods, could lead to an excess intake of some nutrients.
FDA did a thorough review of available data and concluded that “...total nutrient intakes (from both conventional foods and dietary supplements)...do not exceed the [Tolerable Upper Levels] for most vitamins and minerals.”
“In fact, the data show that consumers are not meeting even the EAR for a number of nutrients. We still do urge people, however, to be aware of their total nutrient intake from foods, fortified foods and supplements. But FDA’s analysis should help put to rest those critics using overnutrification as a way to dismiss supplementation,” Wong said.