FDA warns against “misleading” weight loss pills
FDA tests revealed that the capsules contain "excessive amounts of sibutramine," a stimulant which could cause high blood pressure, stroke or heart attack in some consumers. “Sibutramine is not listed among the pill’s ingredients and could harm users, especially those with heart conditions,” according to an FDA warning statement.
Heart palpitations
Even healthy users could experience harmful side effects such as anxiety, nausea, heart palpitations, and increased heart rate or increased blood pressure, it added. In addition sibutramine can interact with other medications to create side effects that are even more hazardous.
A powerful stimulant, sibutramine should not be used without a prescription due to the safety risks associated with it.
FDA said the product’s label is misleading because it “….indicates that it is a natural vitamin and calcium capsule for use in children as young as two years old."
The capsules are sold online by Beautiful Health Inc., formerly known as LL Health and Beauty.
Sample packets are also distributed at community events such as the 40th Annual Mexican Independence Day Parade in Chicago last month.
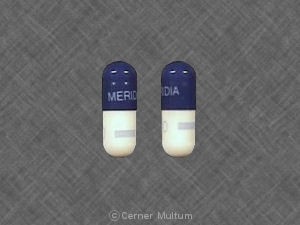
Slimming Beauty pill packets contain four capsules in a small plastic bag with English and Spanish wording.
Side effects
According to Slimming Beauty’s packaging circulated by the FDA, the company claims: “The capsule has passed the Chinese drug certification food GMP (Good Management Practice) standard certification and is proved to be safe and free of side effects.”
But FDA officials have reported several cases of major side effects caused by using the product.
A label on the package headed “Supplement Facts” lists the contents as being: Bitter orange, mulberry leaf, jobstears seed, evening primrose oil, cassia seed, oriental water plantain rhizome and lotus leaf.
Consumers and doctors are asked to report adverse side effects associated with using Slimming Beauty capsules to the FDA at www.fda.gov.medwatch/report.htm.