FDA warns against Venom sibutramine supplements
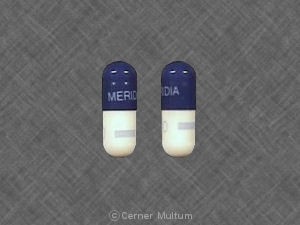
Sibutramine goes under the trade name Meridia in the US and Canada and Reductil in Europe and most other countries and is used as a weight loss agent in pharmaceutical products but is not approved for use in dietary supplements.
“Sibutramine, a controlled substance with risks for abuse or addiction, is a potent drug that poses potential safety risks,” FDA said in its advisory.
“Sibutramine is the active ingredient in an FDA-approved prescription drug used as an appetite suppressant for weight loss,” said Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research.
“But when present in a dietary supplement, it may harm unsuspecting consumers because sibutramine can substantially increase blood pressure and heart rate (pulse), and may present a significant risk for people with a history of heart disease, heart failure, irregular heart beats or stroke.”
Recall
Venom Hyperdrive 3.0 is based on a "herbal blend" and manufactured by Nevada-based Applied Lifescience Research Industries, which initiated a voluntary recall on the products after being informed by the FDA of the problem.
On its website the company said “trace amounts” of sibutramine were found in the Venom product, but FDA lab test showed “significant amounts of sibutramine per dosage unit.”
“Consumers who have this product should stop taking it immediately and contact their health care professional if they have experienced any adverse effects,” FDA said.
It said serious adverse events, side effects or product quality problems could be reported to its Adverse Event Reporting program either online, by regular mail, fax or phone.
Venom sold via US distributors and retail stores as well as in Canada, Poland, Sweden, Hungary, South Africa, the Netherlands, Australia, France and the United Kingdom.